Durable Medical Equipment (DME) is defined as any medical equipment used in the home to aid in a better quality of living. The term includes wheelchairs, knee braces, hospital beds, nebulizers, walkers, etc. DME is a benefit included in most insurances including Medicare Part B which covers medically necessary DME that is prescribed by a physician for use in a patient’s home.
According to an AARP Bulletin titled “Medicare Under Assault From Fraudsters”, “roughly 10 cents of every dollar budgeted for the giant health insurance program is stolen or misdirected before it helps any enrollee. Malcom Sparrow, a Harvard University professor and leading expert on health care fraud, says the true amount lost to fraud, abuse or improper payments could be 20 percent, or even as high as 30 percent.” See below a graphic that puts the scale of Medicare fraud into perspective.
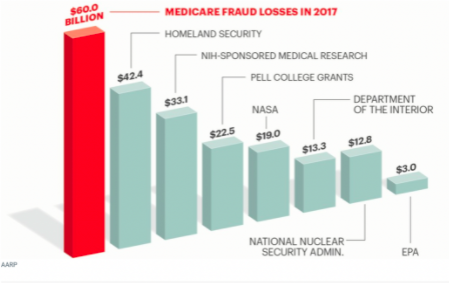
As you can see, DME fraud has become a huge business for criminals, and they are increasingly using MSBs to provide illegal kickbacks to physicians, launder their illegal proceeds, and take advantage of fraud victims. This behavior should disgust any American taxpayer, if not morally, think about how much of our hard-earned money over the years goes straight into these fraudsters’ pockets.
How it works:
See below an excerpt from a February 4, 2021, Department of Justice Press release titled “Florida Businesswoman Pleads Guilty to Criminal Health Care and Tax Fraud Charges and Agrees to $20.3 Million Civil False Claims Act Settlement”
[According to court documents, Wolfe and her conspirators used Regency to establish dozens of DME supply companies — or, rather, DME fronts — using trickery and deception. The scheme involved placing the DME fronts in the names of straw owners. By concealing the true ownership, Wolfe’s conspirators secretly gained control of multiple companies. With such control, they collectively submitted well over $400 million in illegal DME claims to Medicare and the Civilian Health and Medical Program of the VA. The conspirators claimed that the unusually high volume of claims reflected the use of telemedicine procedures, when, in fact, they had simply bribed doctors to approve them. Almost always, the doctors had no telehealth interaction with the beneficiaries.]
As you can see Wolfe established multiple shell companies and submitted over $400 million in illegal DME claims to Medicare and the Civilian Health and Medical Program of the VA. Another crucial element of the scheme is she bribed doctors to approve these fraudulent invoices stating they conducted telehealth services for the beneficiaries. The illegal kickbacks to doctors is what check cashing MSBs will encounter. A pattern we have picked up on is checks from companies with “Medical Equipment, DME, Braces, Devices, etc.” made payable to individuals that turn out to be physicians or money mules are likely illegal kickbacks to doctors for signing off on fraudulent DME claims.
Anti-Kickback Statute [42 U.S.C. § 1320a-7b(b)]
The AKS is a criminal law that prohibits the knowing and willful payment of “remuneration” to induce or reward patient referrals or the generation of business involving any item or service payable by the Federal health care programs (e.g., drugs, supplies, or health care services for Medicare or Medicaid patients). Remuneration includes anything of value and can take many forms besides cash, such as free rent, expensive hotel stays and meals, and excessive compensation for medical directorships or consultancies. In some industries, it is acceptable to reward those who refer business to you. However, in the Federal health care programs, paying for referrals is a crime. The statute covers the payers of kickbacks-those who offer or pay remuneration- as well as the recipients of kickbacks-those who solicit or receive remuneration. Each party’s intent is a key element of their liability under the AKS.
Criminal penalties and administrative sanctions for violating the AKS include fines, jail terms, and exclusion from participation in the Federal health care programs. Under the CMPL, physicians who pay or accept kickbacks also face penalties of up to $50,000 per kickback plus three times the amount of the remuneration.
Other Examples:
- https://www.justice.gov/usao-nj/pr/five-people-charged-two-others-admit-guilt-93-million-health-care-fraud-scheme
- https://www.justice.gov/usao-ednc/pr/north-carolina-durable-medical-equipment-corporation-sentenced-10-million-healthcare
- https://www.justice.gov/usao-ma/pr/two-women-plead-guilty-multi-million-dollar-medicare-fraud-scheme
- https://www.justice.gov/usao-sdga/pr/durable-medical-equipment-company-owner-sentenced-federal-prison-bribery-conspiracy
Red flags we have identified relating to DME fraud kickbacks are as follows:
• Maker names related to medical equipment writing checks out to individuals. The dollar amount can be random, but we see a lot in the $2,000 – $9,000 range. Less sophisticated actors will structure these payments to remain under the $10,000 CTR threshold.
• When you look up the registered agents of these DME companies on publicly available databases such as Sunbiz.org in Florida, they often control several other companies related to medical equipment that have been recently established. This is indicative of shell companies just like we saw in the above example from the DOJ press release.
• A payee that comes into the check cashing store and regularly cashes checks made payable from DME related companies. You may want to conduct and internet search of these individuals and determine if they are a physician.
What to do if you suspect DME fraudsters are taking advantage of your business to facilitate payments?
If you suspect a potential customer or customer is involved in DME fraud you should investigate to determine if any of the above red flags are involved. As always, you should ask yourself does this activity make sense given the nature of the alleged business? If you determine an existing customer is involved in DME fraud and has conducted transactions aggregating over the SAR dollar volume threshold, file a SAR. As always if you suspect a customer is involved in illegal activity you need to block them from doing business with you. No matter how profitable the relationship is, it’s not worth being fined, losing your license, or ending up in a Department of Justice press release.
Sources:
https://www.aarp.org/money/scams-fraud/info-2018/medicare-scams-fraud-identity-theft.html
https://oig.hhs.gov/compliance/physician-education/01laws.asp
Recent Posts
How to Improve Monitoring and Reporting
Better Monitoring Per federal and state guidelines, MSBs...
How to Think About Employee Training
In this article we want to answer the major questions...
Suspicious Activity Reporting
Suspicious Activity Reporting Suspicious activity...




